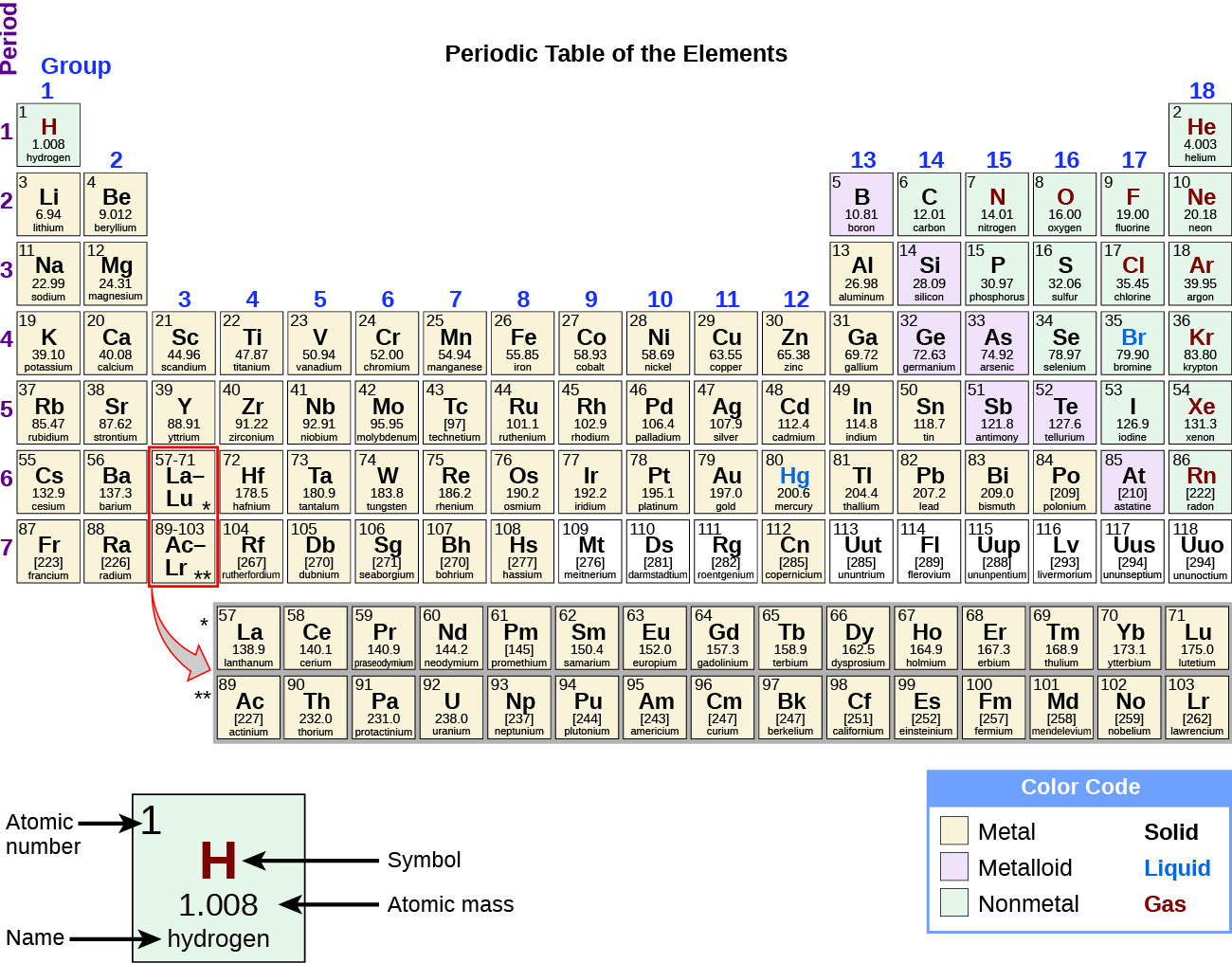
The vertical columns, or groups, indicate elements with similar chemical properties. When the outer shell is full of electrons, the element is a noble gas. As you move across the periodic table, it indicates gradual filling of electron shells. For instance, alkali metals, transition metals, nonmetals, or noble gases D. Often the periodic table is color-coded to indicate similar types of elements. Identify the groups of the periodic table Identify elements as metals, nonmetals, metalloids Infer likely properties of elements based on their position on the periodic table. For instance, the bottom row represents the unreactive noble gases. The horizontal rows of the table, or periods, indicate chemical reactivity. Metals, Metalloids, and Nonmetals Periodic Tables. The position of an element in the periodic table gives you a quick indication of what properties it has and how it is likely to behave. This free periodic table is color-coded to indicate the electronegativity of an atom of an element. 
Of the following statements about the periodic table, which one is INCORRECT? A. Color code states of matter (solid, liquid, gas) at room temperature on the Periodic Table. Color code the metals, nonmetals, and metalloids on the Periodic Table. Color Coding the Periodic Table Atoms & Elements Unit Students will be able to. The scientific community settled on the current one based on Mendeleev's 1869 periodic table, because it is so useful. Our development team has been informed of the issue. They are separated by a diagonal band of semi metals.In the early years, there were actually multiple ways of graphically representing information about the elements. Metals, Nonmetals, Metalloids 422 plays 8th - 12th 15 Qs. Metals and Nonmetals 2.9K plays 6th - 8th 10 Qs. Find other quizzes for and more on Quizizz for free Skip to Content. Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Periodic Table/Metals/Nonmetals quiz for 6th grade students. Semi metals exhibit properties intermediate between those of metals and nonmetals. The elements can be broadly divided into metals, nonmetals, and semi metals. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. 
The periodic table is an arrangement of the elements in order of increasing atomic number. Each group is located in a different part of the periodic table. Often the periodic table is color-coded to indicate similar types of elements. Metals are also malleable (they can be beaten into thin sheets) and ductile (they. For instance, the bottom row represents the unreactive noble gases. A metal is a substance that is shiny, typically (but not always) silvery in color, and an excellent conductor of electricity and heat. Elements are either metals, nonmetals, or metalloids (or semi metals). Every element can be classified as either a metal, a nonmetal, or a metalloid (or semi metal), as shown in Figure 1.11.2 1.11.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
